
CPC #5: Answer
The
patient's presentation is that of
an adult with jaundice. The differential
diagnosis lies between intrahepatic
and extrahepatic causes of cholestasis.
The imaging studies performed clearly
show that this is an extrahepatic
cholestasis syndrome: the ERCP demonstrates
stricture involving the extrahepatic
bile duct and pancreatic duct yielding
the " double duct sign,"
while the CT scan shows a cystic
lesion within the pancreatic tail.
Despite the fact that a discrete
pancreatic mass was not identified
on CT scan, the clinical, laboratory,
and radiologic findings all point
toward pancreatic carcinoma. First,
although somewhat nonspecific, the
highly elevated CA19-9 level (35-40
is the normal range) suggests pancreatic
or bile duct carcinoma. Second,
the patient did not demonstrate
calculi on imaging studies, and
his current area of stricture is
distal to the surgical clips of
his prior cholecystectomy, which
makes stricture secondary to accidental
damage to the bile ducts from the
prior cholecystectomy improbable.
A Klatskin tumor is eliminated by
the fact that the obstruction is
distal in the pancreatic and bile
ducts, while ampullary and duodenal
tumors are excluded by the fact
that they were not visualized on
the ERCP. Hence, the tumor must
be in the pancreatic or distal bile
ducts. The area of stricture of
the pancreatic duct on ERCP is at
the knee of the pancreatic duct,
which is the classic area in which
pancreatic carcinomas arise.
After informed consent was obtained and the patient educated, the patient was explored surgically. He was found to have a mass within the head of the pancreas, and a pylorus preserving pancreatoduodenectomy was performed. Intraoperative examination of the pancreatoduodenectomy specimen revealed infiltrating adenocarcinoma within the head of the pancreas, with extension of the carcinoma to the pancreatic neck surgical margin. This latter finding necessitated conversion of the procedure to a pylorus preserving total pancreatectomy and splenectomy, with creation of a duodenal-jejunal and hepato-duodenal anastamoses. Histopathologic examination of the resection specimen revealed a 1.5 cm infiltrating adenocarcinoma within the head of the pancreas (Figure 1, Figure 2), which had metastasized to 5 of 22 peripancreatic lymph nodes (Figure 3). The tail of the pancreas contained an intraductal papillary mucinous neoplasm (IPMN) (Figure 4), which may have represented a precursor to this patient's invasive carcinoma. The patient did well postoperatively and was discharged home on postoperative day number 8. He is now an insulin diabetic and requires pancreatic enzyme supplementation with his meals. He visited the surgical clinic at six weeks, and had gained weight and looked well. He was scheduled to begin adjuvant chemotherapy and radiation therapy.
Figure
1: 1.5 cm infiltrating adenocarcinoma
within the head of the pancreas

Figure
2: Infiltrating pancreatic adenocarcinoma

Figure
3:
metastasis to peripancreatic lymph
node

Figure
4: The tail of the pancreas
contained an intraductal papillary
mucinous neoplasm (IPMN) which
may have represented a precursor
to this patient's invasive carcinoma.
Discussion:
Causes of jaundice can be divided into prehepatic, hepatic and post hepatic categories. Prehepatic jaundice typically results from hemolysis and is evident on clinical and laboratory studies. For example, heomolytic disease of the newborn (erythroblastosis fetalis) can lead to significant hyperbilirubinemia which can, in turn, cause significant brain damage. Intrahepatic causes of jaundice include viral hepatitis, cholestatic drug reaction, alcohol toxicity, cirrhosis, and the inherited Gilbert's syndrome. Extrahepatic causes of cholestasis include gallstones, obstructing tumors, benign strictures such as one sees secondary to pancreatitis and status post cholecystectomy when the bile duct is injured, and congenital abnormalities such as choledochal cysts, Caroli's disease and primary sclerosing cholangitis. In general, the serum bilirubin must be greater than 3 mg per deciliter in order for one to appreciate clinical jaundice in a Caucasian patient's sclera.
The key radiographic feature, which helps in the distinction between extrahepatic and intrahepatic cholestasis, is the presence of dilated bile and pancreatic ducts. The absence of dilated pancreatic and biliary ducts suggests intrahepatic cholestasis, and necessitates a medical work-up including hepatitis serologies, and perhaps percutaneous liver biopsy. The presence of dilated ducts generally indicates surgical disease. One can demonstrate the presence of dilated ducts using either a right upper quadrant ultrasound or a CT scan. One should generally order one or the other of these tests but not both. In general, one should order the right upper quadrant ultrasound when the patient presents with symptoms consistent with gallstones. The usual scenario is a young patient with symptoms such as flatulence and belching who does not show weight loss. The right upper quadrant ultrasound is very helpful for demonstrating dilated bile ducts, demonstrating the presence of gallstones in the gallbladder or even gallstones within the bile duct. It is not helpful for delineating carcinomas in the ampulla, as there is too much duodenal air to allow masses in this region to be easily discerned. CT scan is generally indicated in older patients with weight loss in whom cancer is the major differential. The CT scan is useful because it not only demonstrates the presence of dilated ducts, but also allows one to image the periampullary area. One may actually stage periampullary tumors by the presence of lymphadenopathy, serosal implants or involvement of mesenteric vessels that will be evident on CT scan. If further imaging studies are needed to delineate a tumor, ERCP and Percutaneous Transhepatic Cholangiogram (PTC) can be used. In general, ERCP is useful for ampullary tumors, whereas PTC is useful for proximal bile duct tumors that obstruct the confluence of the bile ducts.
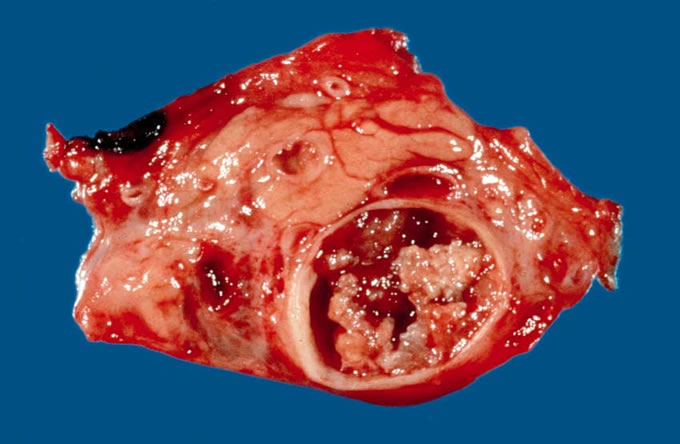
From a pathologic point of view, a variety of lesions may be cystic within the pancreas (Figure 5). Pancreatic pseudocyst is perhaps the most common. It is called a pseudocyst because it lacks a true epithelial lining, and is typically seen in association with pancreatitis. Serous cystadenomas are characterized by a circumscribed, sponge-like gross appearance with a central stellate scar (Figure 6). They are characterized by clear glycogen-rich cells which lack cytologic atypia (Figure 7). These lesions have an almost uniformly benign course. Solid-cystic-pseudopapillary tumor typically affects young females. It is characterized by uniform polygonal cells which cling to blood vessels and are associated with macrophages. This lesion again usually has a benign course. Mucinous cystic neoplasms of the pancreas are characterized by their circumscription, predominance in females, and typical location in the pancreatic tail. Under the microscope, these lesions have ovarian type stroma and a mucinous lining which ranges from bland to carcinoma in situ. In one-third of cases, mucinous cystic neoplasms have an associated invasive carcinoma. The final cystic lesion is the intraductal papillary mucinous neoplasm. This lesion grossly involves the main pancreatic duct (Figure 8), and may present with gross mucinous extravasation out of the pancreatic duct on ERCP examination. Under the microscope, the markedly dilated pancreatic ducts are lined by papillary mucinous epithelium which, demonstrates a range of cytologic atypia ranging from benign to borderline to carcinoma in situ with invasive carcinoma representing a potential complication (Figure 9 ).
This patient's clinical course demonstrates the challenges that pancreatic cancer, an almost uniformly fatal neoplasm today, poses to treating physicians. This patient clearly had symptoms nine months earlier when he had his gallbladder removed. However, a pancreatic mass was not identified, even though it seems more than likely that his IPMN was present at that time. Even though this patient's invasive carcinoma was caught at a relatively small size of 1.5 cm., he already had significant nodal metastases at the time of diagnosis, and his overall prognosis is now guarded. The goal of our research today is to detect such precursor lesions at an early stage when they are treatable. Along these lines, new markers of pancreatic cancer are rapidly being discovered using the wealth of genetic data generated through the Human Genome Project. It is hoped that these novel markers will lead to blood-based screening tests which could have helped patients such as this. Catching this lethal cancer early appears to be the best hope for obtaining a cure.
Figure
5 : From a pathologic point
of view, a variety of lesions may
be cystic within the pancreas.

Figure
6: Serous cystadenomas are characterized
by a circumscribed, sponge-like
gross appearance with a central
stellate scar.
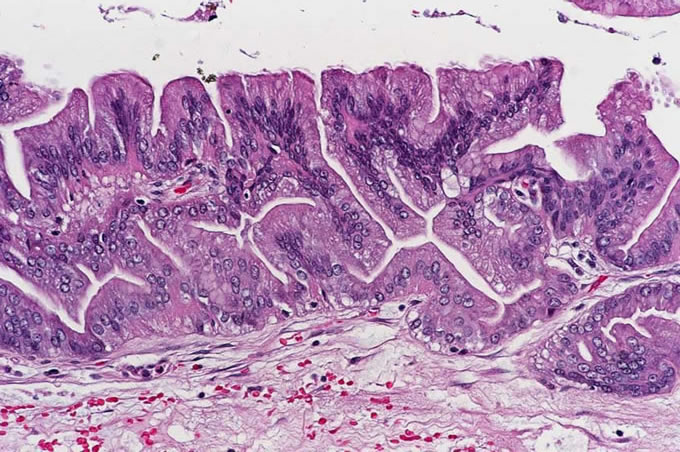
Figure
7: Serous cystadenomas are characterized
by clear glycogen-rich cells which
lack cytologic atypia.

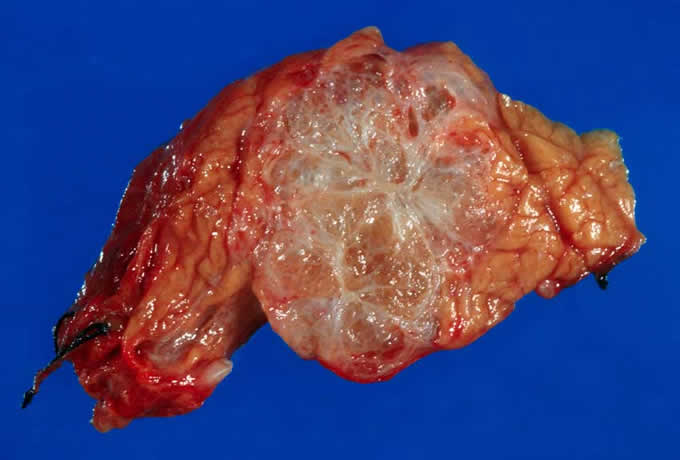
Figure
8: This intraductal pancreatic
mucinous neoplasm (IPMN) grossly
involves the main pancreatic ducts.
Figure
9: Under the microscope, the
markedy dilated pancreatic ducts
are lined by atypical papillary
mucinous epithelium (left), which
develops into invasive carcinoma
(right).
